
![]()
![]()
![]()
![]()
1 Sandra Bošković
1 Deana Švaljug
1 Marija Spevan
1 Andrica Lekić
1 Iva Keglević
1 Silvije Šegulja
![]()
![]()
![]()
![]()
Sandra Bošković
University of Rijeka, Faculty of Health Studies, Rijeka, Croatia
E-mail: sandra.boskovic@uniri.hr
![]()
![]()
![]()
+ 6/7 weeks of gestation, and assessed as healthy during first initial clinical examination. Pain was as- sessed during routine blood sampling for phenylke- tonuria screening, and assessments were performed by trained nurses. Statistical analysis used descrip- tive and inferential methods (Wilcoxon test, Fried- man’s ANOVA, Cochran’s Q test) and Cronbach’s alpha coefficient.
Results. The Croatian NIPS version showed high in- ternal consistency (Cronbach’s α = 0.846). Significant physiological and behavioral responses were record- ed before (mean 1.51), during (mean 6.66) and after (mean 2.02) the painful procedure (p < 0.001).
![]()
![]()
![]()
The most common methods of pain assessment in pediatrics are self-assessment (not applicable in the neonatal period), behavioral assessment, and meas- urement of physiological parameters (1). Behavioral indicators include irritability, tremors, crying, grimac- ing, body movements, but also pathological calmness, lethargy, sleep disturbances, and loss of appetite. Physiological parameters encompass changes in skin color, variations in heart rate, respiratory rate, blood pressure, and neuroendocrine responses (e.g., corti- sol and growth hormone levels) (2). The most reliable assessment is based on a combination of behavioral and physiological indicators, although in practice it is challenging to balance reliability with the time required for assessment (3). Despite its importance, standardized pain assessment scales for newborns are still not used in neonatal care in Croatia.
A review of the available neonatal pain assessment scales identified the NIPS (4) as a particularly valid and reliable instrument, a finding supported by numerous international adaptations and validation studies. Ex- amination of the evidence confirms that the Neona- tal Infant Pain Scale is a multidimensional tool with robust psychometric properties, including excellent inter-rater reliability, concurrent validity, construct validity, and predictive validity—features that are es- sential for accurate pain assessment and for guiding appropriate clinical interventions, especially in com- parison with other pain assessment instruments. Fur- thermore, multiple studies demonstrate the success- ful implementation of NIPS and highlight its ease of use in routine clinical practice (5-7).
Therefore, as part of the scientific project uniri-iskus- ni-biomed-23-76, the implementation of systematic procedures for pain assessment and relief is planned as a standard in neonatal care. Special emphasis is placed on introducing the Neonatal Infant Pain Scale (NIPS), which assesses facial expression, crying, breathing patterns, arm and leg movements, and state of arousal (8). In combination with monitoring vital pa- rameters, this scale will enable reliable assessment of pain intensity during medical procedures. This will cre- ate the prerequisites for the systematic introduction of all procedures for the prevention, relief, and treat- ment of pain in newborns in the Republic of Croatia.
![]()
The aim of this study is to adapt Neonatal Infant Pain Scale (NIPS) instrument for pain assessment in new- borns, in order to evaluate its content validity and reliability, with the goal to introduce and apply the scale in neonatal care in Croatia.
![]()
![]()
Data were collected at the Clinical Hospital Centre Ri- jeka between October 2024 and January 2025. A to- tal of 94 healthy newborns, delivered either vaginal- ly or by cesarean section, participated in the study. Before the pain assessment procedure, both parents read and signed informed consent and voluntarily agreed to the participation of their newborn in the study. During the study period at the Clinical Hospital Centre Rijeka, a total of 534 healthy newborns were delivered, which represents an 18% response rate.
After the back-translated NIPS had been approved by the author of the original instrument, a panel of ex- pert judges assessed the cross-cultural equivalence between the original and the adapted versions. All modifications made during the translation process resulted in a preliminary version of the NIPS. The expert panel consisted of a professor with exper- tise in cross-cultural adaptation, a pain management specialist, and a language professional. Each expert received the original instrument, the synthesized forward and back-translated versions, as well as all comments provided by the translators and research- ers throughout the adaptation process.
The panel systematically compared all versions of the instrument and evaluated idiomatic, experien- tial, conceptual, and semantic equivalence. Semantic equivalence was assessed by classifying each item as having “exactly the same meaning,” “nearly the same meaning,” or “a different meaning” compared with the corresponding item in the original scale. Following this evaluation and consensus discussion, a preliminary final version of the Croatian NIPS was established.
In the second phase of the study, the reliability, and clinical application of the instrument were assessed. Data were collected with the help of three nurses employed at the Clinical Hospital Centre Rijeka, who were asked to apply the scale to a sample of newborns. Prior to using the scale, two researchers trained the nurses on how to apply the scale, assess parameters, and record data in the questionnaire. The sample consisted of newborns undergoing rou- tine blood sampling as part of the National Newborn Screening Program of the Newborn Screening Com- mittee of the Ministry of Health of the Republic of Croatia. Screening is conducted as an organized sys- tem of testing for certain congenital diseases in all newborns of a defined population, with the aim of identifying them before they cause adverse health outcomes. As in most countries, newborn screening is a mandatory health protection measure in Croatia. Blood samples for screening are collected between the third and fifth day of life, and no later than the eighth day. Blood is most often drawn from the new- born’s heel while still in the maternity ward (9). The inclusion criteria for newborns in the study were: gestational age from 37 + 0/7 weeks to 41 + 6/7 weeks; newborns classified as healthy according to their first clinical examination; Apgar scores of ≥7 at one and five minutes, as lower scores may be as- sociated with alterations in central nervous system pain-processing mechanisms (10). Both vaginally and caesarean-delivered newborns were included, as the study aimed to assess whether there is a differ- ence in pain perception between these two modes of delivery. In addition, the following exclusion cri- teria were applied: maternal use of opioids, as these substances may cross the placental barrier and cause changes in neonatal nociceptive pathways; maternal use of alcohol or drugs; mother under 18 years of age without a legal guardian present; mother with vertically transmittable infectious diseases such as syphilis, toxoplasmosis, cytomegalovirus infection, mumps, herpes, hepatitis B, and HIV/AIDS; and new- borns with visible congenital malformations (9).
The Neonatal Infant Pain Scale (NIPS) (4) is a behav- ioral scale used for pain assessment. It is designed to evaluate procedural pain in newborns. This scale takes into account pain assessment during and after a painful procedure and considers several behavio- ral indicators for assessing pain in both preterm and
full-term newborns. The scale can be used to monitor a newborn before, during, and after a painful proce- dure, such as venipuncture. The instrument was de- veloped at the Children’s Hospital of Eastern Ontario.
The assessment parameters include:
Facial expression (Q1): relaxed muscles, neu- tral calm face, grimaces (tense facial muscles, furrowed brows, chin/jaw).
Crying (Q2): none, whimpering (mild intermittent moaning), and vigorous cry (loud screaming).
Breathing patterns (Q3): relaxed breathing, changes in breathing.
Arm and leg movements (Q4 and Q5): relaxed/ restrained (no muscle stiffness, occasional ran- dom limb movements), flexion/extension (tense, straight, rigid, and/or rapid extension or flexion).
State of arousal (Q6): sleep/awake (quiet, calm sleep or alert and calm), restless/nervous (awake, restless).
Each parameter is scored from 0 to 2 points, with a total score of 0–2 indicating no pain, 3–4 indicating moderate pain, and a score greater than 4 indicating severe pain (4).
The study protocol was approved by the Ethics Com- mittee of the Clinical Hospital Center Rijeka and the Ethics Committee for Biomedical Research of the Faculty of Health Studies, University of Rijeka (Class: 003-05/20-1/08, Reg. No.: 2170-29-02/1-20-02).
Written informed consent for participation was ob- tained from the parents of all children after they had received both oral and written information regarding the study objectives, voluntary participation, ethical considerations, and data protection. The study was conducted in accordance with the principles of the Declaration of Helsinki.
Statistical analyses were performed using Statistica, version 14.1.0.8 (Cloud Software Inc.). Statistical sig- nificance was set at p ≤ 0.05, with 95% confidence intervals. The sample size was determined based on the rule of thumb recommending 10 cases per item. Since the instrument contained six items, a minimum sample size of approximately 60 participants was required. A power/precision calculation with 80%
power and a 5% margin of error indicated a required sample size of 89 participants. Descriptive statistics were used to summarize the data, including mean and standard deviation. For nominal variables, frequencies and percentages were reported. The Kolmogorov- Smirnov test was applied to assess the normality of the data distribution. Since the data were not normally distributed, non-parametric tests were used. The Wil- coxon matched-pairs test was applied for two depend- ent samples, and Friedman ANOVA for three groups.
Repeated-measures ANOVA was performed for the vital parameters because the data are quantitative, and the Kolmogorov–Smirnov test indicated that they follow a normal distribution. The NIPS questionnaire, on the other hand, yields ordinal data; therefore, a nonparametric ANOVA was used to examine differ- ences in the total score of the six behavioral indica- tors before, during, and after the painful procedure.
For categorical data with three groups, Cochran’s Q test was employed. The reliability of the question- naires was assessed using Cronbach’s alpha coeffi- cients, as well as by calculating the change in Cron- bach’s alpha if individual items were removed from the respective scale.
![]()
![]()
The study included a total of 94 healthy newborns delivered via vaginal or cesarean section. General characteristics for the participants are summarized in Table 1. A total of 79.8% of the infants were born at 37+0/7 weeks of gestation, 4.2% at 37 weeks, and 16% after 38 weeks. Apgar scores were recorded at two intervals, with the second measurement being significantly higher than the first (p < 0.001).
To compare heart rate and oxygen saturation before, during, and after the painful procedure, repeated measures ANOVA (RM ANOVA) with Bonferroni post- hoc testing was applied. The results are presented in Table 2.
Table 2. Heart rate and oxygen saturation before, during, and after the painful procedure | |||
Mean | 95% CI | p | |
Pulse before | 124.18 | 121.34– 127.02 | |
Pulse during | 144.15 | 140.10– 148.20 | < 0.001 |
Puls after | 148.02 | 143.58– 152.45 | |
oxygenation before | 98.79 | 98.59– 99.01 | |
oxygenation during | 97.55 | 97.18– 97.92 | < 0.001 |
oxygenation after | 97.83 | 97.48– 98.17 | |
Note: 95% CI- confidence interval, p - statistical significance | |||
Heart rate and oxygen saturation differed signifi- cantly between the pre- and intra-procedure meas- urements, as well as between the pre- and post- procedure measurements (p < 0.001). Changes in respiratory rate were significantly more frequent during the painful procedure (p < 0.01).
Facial expression (Q1) revealed that 22.4% of new- borns grimaced before the procedure, compared to 24.5% after the procedure. During the painful proce- dure, all newborns exhibited grimacing. Crying (Q2)
Table 1. General characteristics (N = 94) | |||||
N = 94 | Mean | Min | Max | SD | Z* p |
Apgar 1 | 9.78 | 7 | 10 | 0.59 | |
Apgar 2 | 9.98 | 9 | 10 | 0.14 | |
Mass /g | 3476.92 | 2240 | 4330 | 481.88 | |
Body length/cm | 50.07 | 43 | 55 | 2.36 | |
Note: N- number SD- standard deviation *Wilcoxon matched-pairs test p - statistical significance | |||||
3.3 0.001
showed significant differences across all time points: 27.7% of newborns cried before the procedure, com- pared to 51.1% after.
Significant differences were observed across all be- havioral assessment parameters (Q1–Q6) when com- paring responses before, during, and after the painful procedure (Table 3.)
Figure 1. shows the total score of the six behavio- ral indicators before, during, and after the painful procedure.
The Friedman ANOVA revealed a significant overall difference (p < 0.001). Post-hoc analyses indicated significant differences between the pre-procedure and during-procedure measurements, as well as between the during-procedure and post-procedure measurements. No significant difference was ob- served between the pre- and post-procedure meas- urements (p = 0.068).
![]()
![]()
The aim of this study was to adapt the Neonatal Infant Pain Scale (NIPS) for the Croatian-speaking area and to evaluate its reliability. The results dem- onstrate high internal consistency of the scale (α = 0.768–0.891), which is consistent with previous stud- ies by Lawrence et al. (4) and Hudson-Barr (11). Al- though the inter-rater agreement (ICC 0.516–0.759) was moderate and somewhat lower compared to the excellent results reported by Sarhangi et al. (6), the
presence of parents suggests that situational factors may influence the intensity of the response captured by the scale, further emphasizing the importance of standardized assessment conditions.
The decision to validate the NIPS scale in Croatia (Bošković and Ličen (1)) is based on its successful cul- tural adaptation in numerous countries (5). The find- ings of this research provide an empirical basis for its integration into neonatal care standards in Croatia.
The primary limitation of this study is the relatively small sample size, which restricts the generalizability of the results to the broader national population and various gestational ages. Additionally, the research focused primarily on reliability and internal consist- ency, while aspects such as concurrent validity in re- lation to other behavioral scales within the Croatian context remain less explored.
Future research should focus on validating the scale using a larger sample to achieve full national stand- ardization. Comparative studies involving multiple pain assessment scales are recommended to identify the most sensitive instrument for specific types of stimuli (e.g., postoperative vs. procedural pain). Fur- thermore, it is necessary to investigate the impact of systematic NIPS implementation on long-term clini- cal outcomes and the effectiveness of pain manage- ment within neonatal intensive care units in Croatia.
![]()
findings confirm that the scale is a stable instrument for application within the local clinical context.
The interpretation of the results indicates a sig- nificant sensitivity of the NIPS scale to behavioral changes. The statistically significant increase in fa- cial grimacing and changes in respiratory patterns during the painful procedure (p < 0.001) confirms the validity of the behavioral indicators measured by the scale. Furthermore, the correlation between the scale scores and physiological deviations (heart rate and oxygen saturation) supports the multidimen- sional nature of the neonatal pain response, aligning with international findings (10 – 12). The observation that a smaller proportion of newborns cried in the
![]()
The Croatian version of the NIPS scale demonstrat- ed good reliability and high internal consistency (α
= 0.846). Significant changes in vital parameters and behavioral indicators (p < 0.001) confirm the instrument’s sensitivity in detecting neonatal pain. This study confirms the psychometric reliability of the scale for clinical use, while further research on a larger sample will enable its full standardization in accordance with national methodological guidelines.
Table 3. Behavioral assessment parameters | |||||
0 | 1 | 2 | p | ||
Before | 73 | 21 | |||
Facial Expression | During | 0 | 94 | < 0.001 | |
After | 71 | 23 | |||
Before | 68 | 26 | 0 | ||
Cry | During | 0 | 27 | 67 | < 0.001 |
After | 46 | 48 | 0 | ||
Before | 76 | 18 | |||
Breathing Patterns | During | 1 | 93 | < 0.001 | |
After | 72 | 22 | |||
Before | 69 | 25 | |||
Arms | During | 3 | 91 | < 0.001 | |
After | 60 | 34 | |||
Before | 70 | 24 | |||
Legs | During | 1 | 93 | < 0.001 | |
After | 66 | 28 | |||
Before | 66 | 28 | |||
State of Arousal | During | 0 | 94 | < 0.001 | |
After | 59 | 35 | |||
Note: p - statistical significance | |||||
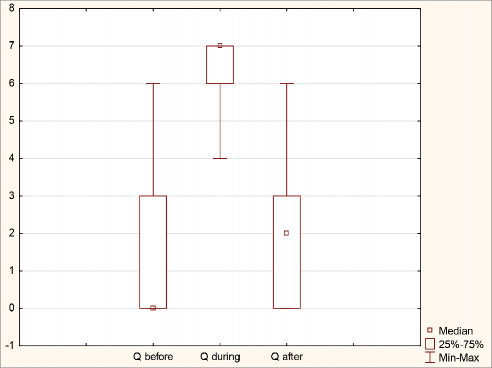
Figure 1. Behavioral Responses
Conceptualization and methodology (SB, MS, DŠ, IK, SŠ, AL); data curation and formal analysis (SB, MS, DŠ, IK, SŠ, AL); investigation and project administration (SB, MS, DŠ, IK., SŠ, AL); writing – original draft and review & editing (SB, MS, DŠ, IK, SŠ, AL). All authors have approved the final manuscript.
The authors declare no conflicts of interest.
Not applicable.
This research was carried out as part of the scientific project uniri-iskusni-biomed-23-76 of the University of Rijeka. This research did not receive any specific grant from funding agencies in the public, commer- cial, or not-for-profit sectors.
During preparation, the author(s) used ChatGPT for language enhancement.
![]()
![]()
Bošković S, Ličen S. Identification of neonatal infant pain assessment tools as a possibility of their appli- cation in clinical practice in Croatia: an integrative lite- rature review. Pain Manag Nurs. 2021;22(5):674–80. https://doi.org/10.1016/j.pmn.2021.01.003
Fitzgerald M. The development of nociceptive circu- its. Nat Rev Neurosci. 2005;6:507–20. https://doi. org/10.1038/nrn1701
Brummelte S, Grunau RE, Chau V, Poskitt KJ, Brant R, Vinall J, et al. Procedural pain and brain development in premature newborns. Ann Neurol. 2012;71(3):385–
96. https://doi.org/10.1002/ana.22267
Lawrence J, Alcock D, McGrath P, Kay J, MacMurray SB, Dulberg C. The development of a tool to assess neo- natal pain. Neonatal Netw. 1993;12(6):59–66. https:// doi.org/10.1016/0885-3924(91)91127-U
Obiedat H, Al-Maaitah EI. Critique of the use of Neo- natal Infant Pain Scale (NIPS). Neonatal Pediatr Med. 2020;6(1). Available at: https://www.omicsonline.org/ open-access-pdfs/critique-of-the-use-of-neonatal-in- fant-pain-scale-nips.pdf.
Sarhangi F, Mollahadi M, Ebadi A, Matinzadeh ZK, Matinzadeh ZK, Tadrisi SD. Validity and reliability of Neonatal Infant Pain Scale in neonatal intensive care units in Iran. Pak J Med Sci. 2010;27:1087–91. Available at: https://www.researchgate.net/publica- tion/286809287_Validity_and_reliability_of_Neo- natal_Infant_Pain_Scale_in_Neonatal_Intensive_Ca- re_Units_in_Iran_2010
Bellieni C, Cordelli DM, Caliani C, Palazzi C, Franci N, Perrone S, et al. Inter-observer reliability of two pain scales for newborns. Early Hum Dev. 2007;83:549–52. https://doi.org/10.1016/j.earlhumdev.2006.10.006
Lajović J. Localization of measurement instruments: example of the AMS questionnaire. Slovenian Med J. 2008;77(6–7):461-5. Available at: https://vestnik.szd. si/index.php/ZdravVest/article/view/489
Begović A. Kriteriji za provođenje drugostupanjskih testova u novorođenačkom probiru na homocistinuriju i nedostatak vitamina B12 [Master’s thesis]. Zagreb: Sveučilište u Zagrebu, Farmaceutsko-biokemijski fa- kultet; 2023. [Croatian] Available at: https://repozito- rij.pharma.unizg.hr/object/pharma:2796
da Motta GDC, Schardosim JM, da Cunha MLC. Neonatal in- fant pain scale: cross-cultural adaptation and validation in Brazil. J Pain Symptom Manage. 2015;50(3):394–401. https://doi.org/10.1016/j.jpainsymman.2015.03.019
Hudson-Barr D, Capper-Michel B, Lambert S, Palermo TM, Morbeto K, Lombardo S. Validation of the Pain Asse- ssment in Neonates (PAIN) scale with the Neonatal In- fant Pain Scale (NIPS). Neonatal Netw. 2002;21(6):15–
21. https://doi.org/10.1891/0730-0832.21.6.15
Pillai Riddell RR, Bucsea O, Shiff I, Chow C, Gennis HG, Badovinac S, et al. Nonpharmacological management of infant and young child procedural pain. Cochrane Database Syst Rev. 2023;14(6):CD006275. https://doi. org/10.1002/14651858.CD006275